 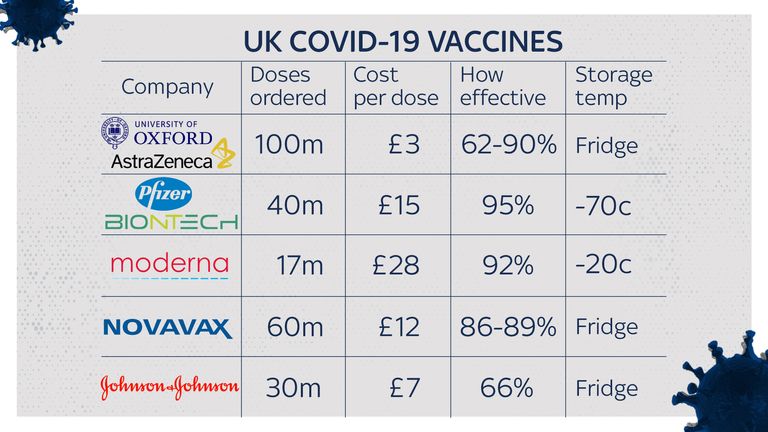
The Moderna COVID-19 Vaccine is administered as a primary series of two doses, one month apart, to individuals 6 months through 17 years of age. “Those trusted with the care of children can have confidence in the safety and effectiveness of these COVID-19 vaccines and can be assured that the agency was thorough in its evaluation of the data.” As we have seen with older age groups, we expect that the vaccines for younger children will provide protection from the most severe outcomes of COVID-19, such as hospitalization and death,” said FDA Commissioner Robert M. “Many parents, caregivers and clinicians have been waiting for a vaccine for younger children and this action will help protect those down to 6 months of age. Prior to making the decision to authorize these vaccines for the respective pediatric populations, the FDA’s independent Vaccines and Related Biological Products Advisory Committee was consulted and voted in support of the authorizations.The agency determined that the known and potential benefits of the Moderna and Pfizer-BioNTech COVID-19 vaccines outweigh the known and potential risks in the pediatric populations authorized for use for each vaccine.The FDA’s evaluation and analysis of the safety, effectiveness and manufacturing data of these vaccines was rigorous and comprehensive, supporting the EUAs. 
The vaccine had been authorized for use in individuals 5 years of age and older.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
